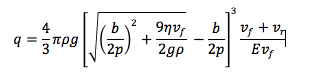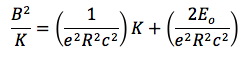In this experiment I calculated the electric charge of an oil drop by measuring the force it experienced from a known electric field. I also calculated how the force, on a charged particle, varied when the electric field changed. Since the force exerted by an electric field on an object with very little charge is also very small, I had to observe tiny oil drops falling and rising really small distances. The main result of this experiment was that focusing on tiny dots for more than 15 minutes made my vision blurry.
The experiment consisted of a two plate capacitator that had a plastic spacer between them. The capacitator was placed within a chamber and had a small hole in the middle where I could introduce the oil drops . There was also a viewing scope mounted so that I could see the hole in the spacer and observe the falling oil drops. I was able to see the drops thanks to a halogen lamp that illuminated the drops. The viewing scope had a grid, whose major lines were separated by 0.5 mm, that allowed me to measure the distance the drops fell.
The capacitators were given voltage by a high voltage DC power supply. There was also a thermistor mounted on the bottom capacitator plate. A thermistor is a resistor whose resistance varies a lot with temperature, and I measured its resistance with a multimeter. With the given resistance I was able to calculate the temperature within the chamber, which was important for later calculations. Lastly, there was an ionization source within the chamber that could be turned on and off. This would create excess electrons that attached themselves to some of the oil drops, and increased their electric charge.
I observed the fall and rise of oil drops in the chamber because the force they experienced also affected their velocities. The force from the electric field would speed up the drops fall and rise. On the other hand, gravity slowed the rise of the particles, so the oil drops velocity was different if it was falling or rising. It seems a little crazy that oil drops can rise, but that was possible because I could reverse the polarization of the electric field. When I did this, I made the electric field and the force point upwards.
It took me forever, but I was able to choose a single oil drop and make it fall and rise in different electric fields, caused by different voltages. I measured the time it took to fall and rise a distance of 0.5 mm, with a voltage of 306.5v, 397v, 483v, 206.2v and 103.1v. I observed that the greater the voltage was, the smaller the time interval was.
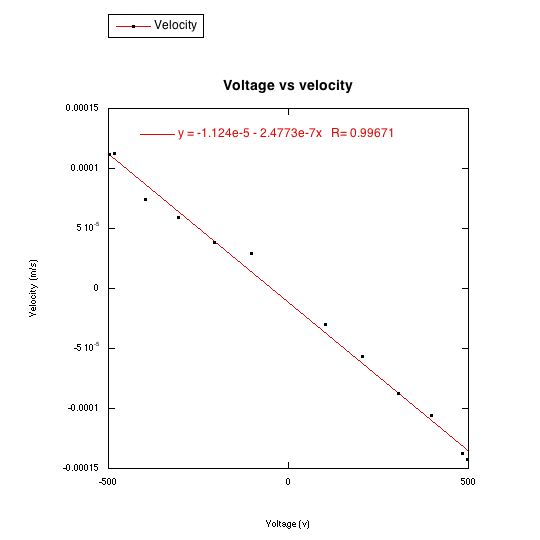
The velocity of falling and rising drops in different voltages increases linearly with voltage. Note that the graph is not centered at the origin.
I also measured the fall and rise of a drop that was exposed to the ionization source. I did not change the electric field and kept the voltage at 245.7 volts. First, I measured the velocity of the drop without turning on the ionization source. Then, I turned on the ionization source for 5 s, and measured the velocity of the drop. I turned on the source one last time and measure the velocity of the drop again. When I turned on the source, it emitted excess electrons that attached themselves to the drops. This increased the charge of the drop and as a result increased the force and the velocity of the drop. This occurred in the first time I turned on the source, but the second time the drop slowed down considerably. Something happened so that the drop didn’t gain electrons, but lost them.

The drop was exposed to an ionization source for 5 second intervals. Curiously, the second exposure resulted in the drop losing electrons instead of gaining them.
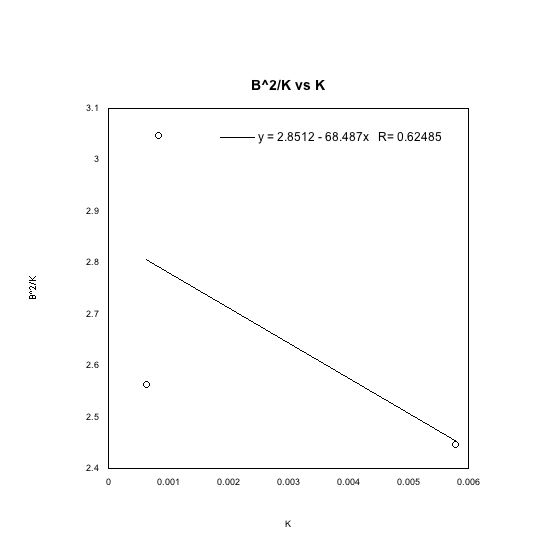
I was able to calculate the electric charge of each drop whose velocity I measured, as well as the drop whose charge changed. I did this with the following equation:
I calculated the charge of each drop, divided it with the charge of the electron and rounded up the result. The drop I whose velocity I measured as function of voltage had six electrons. The drop that wasn’t exposed to the source in the field created by a voltage of 2457 v had 14 electrons. The drop then gained 4 electrons, and had a charge of q=18 e. The drop was exposed a second time to the source and it lost 10 electrons, so it had a charge of q=8e. The loss of electrons cannot be explained as the result of the ionization source, but it is possible another drop stole the electrons. There might be another, better explanation.
In conclusion, the Millikan oil drop experiment is a very pretty experiment and the fact that you can measure the charge of the electron so precisely is kind of amazing. On the other hand, it was very difficult to NOT to lose a drop, and it took hours to find a drop that was a “perfect match” (Wenqi’s words). So, I’m not sure whether I like this experiment or not.